Advertisements
Advertisements
प्रश्न
Match the type of unit cell given in Column I with the features given in Column II.
| Column I | Column II |
| (i) Primitive cubic unit cell | (a) Each of the three perpendicular edges compulsorily have the different edge length i.e; a ≠ b ≠ c. |
| (ii) Body centred cubic unit cell | (b) Number of atoms per unit cell is one. |
| (iii) Face centred cubic unit cell | (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. |
| (iv) End centred orthorhombic cell | (d) In addition to the contribution from unit cell the corner atoms the number of atoms present in a unit cell is one. |
| (e) In addition to the contribution from the corner atoms the number of atoms present in a unit cell is three. |
उत्तर
| Column I | Column II |
| (i) Primitive cubic unit cell | (b) Number of atoms per unit cell is one. |
| (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. | |
| (ii) Body-centred cubic unit cell | (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. |
| (d) In addition to the contribution from unit cell the corner atoms the number of atoms present in a unit cell is one. | |
| (iii) Face centred cubic unit cell | (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. |
| (e) In addition to the contribution from the corner atoms the number of atoms present in a unit cell is three. | |
| (iv) End-centred orthorhombic cell | (a) Each of the three perpendicular edges compulsorily have the different edge length i.e; a ≠ b ≠ c. |
| (d) In addition to the contribution from unit cell the corner atoms the number of atoms present in a unit cell is one. |
Explanation:
(i) 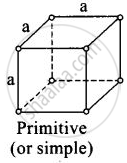
For primitive unit cell, a = b = c
Total number of atoms per unit cell = `1/8 xx 8` = 1
Here, `1/8` is due to contribution of each atom present at comer.
(ii)
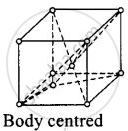
For body centered cubic unit cell, a = b = c.
This lattice contains atoms at comer as well as body centre.
Contribution due to atoms at comer = `1/8 xx 8` = 1
Contribution due to atoms at body centre = 8
(iii)

For face centred unit cell, a = b = c
Total constitutent ions per cell present at corners = `1/8 xx 8` = 1
Total constitutent ions per unit cell present at face centre = `1/2 xx 6` = 3
(iv)
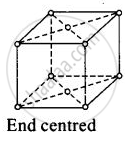
For end centred orthorhombic unit cell, a ≠ b ≠ c
Total contribution of atoms present at corner = `1/8 xx 8` = 1
Total constibution of atoms present at end centre = `1/2 xx 2` = 1
APPEARS IN
संबंधित प्रश्न
Explain how much portion of an atom located at (i) corner and (ii) body-centre of a cubic unit cell is part of its neighbouring unit cell.
An element with molar mass 2.7 × 10-2 kg mol-1 forms a cubic unit cell with edge length 405 pm. If its density is 2.7 × 103 kg m−3, what is the nature of the cubic unit cell?
How can you determine the atomic mass of an unknown metal if you know its density and the dimension of its unit cell? Explain.
An element has atomic mass 93 g mol–1 and density 11.5 g cm–3. If the edge length of its unit cell is 300 pm, identify the type of unit cell
What is the total number of atoms per unit cell in a face-centered cubic structure?
The number of atoms per unit cell in a body centered cubic structure is ____________.
Edge length of unit cell of chromium metal is 287 pm with a bcc arrangement. The atomic radius is of the order:
An element with atomic mass 100 has a bcc structure and edge length 400 pm. The density of element is:
The correct set of quantum numbers for 3d subshell is
The correct set of quantum numbers for 2p sub shell is:
