Advertisements
Advertisements
प्रश्न
Mention any three methods of preparation of haloalkanes from alcohols.
उत्तर
Haloalkanes are prepared by the following methods.
From alcohols: Alcohol can be converted into halo alkenes by reacting it with any one of the following reagents.
- Hydrogen halide
- Phosphorous halides
- Thionyl chloride.
- Reaction with hydrogen halide:
\[\ce{\underset{\text{Ethanol}}{CH3CH2}OH + HCl ->[Anhydrous ZnCl2][\Delta] \underset{\text{Chloroethane}}{CH3CH2Cl + H2 O}}\]
Mixture of con. HCl and anhydrous ZnCl2 is called Lucas Reagent.
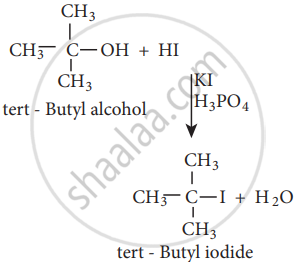
The order of reactivity of halo acids with alcohol is in the order HI > HBr > HCl.
The order of reactivity of alcohols with halo acid is tertiary > secondary > primary.
- Reaction with phosphorous halides:
Alcohols react with PX5 or PX3 to form haloalkanes.
Example:
\[\ce{\underset{\text{Ethane}}{CH3CH2}OH + PCl5 ->\underset{\text{Chloro ethane}}{CH3CH2Cl +}POCl3 + HCl}\]
\[\ce{\underset{\text{Ethanol}}{3CH3CH2OH} + PCl3 -> \underset{\text{Chloro ethane}}{3 CH3CH2Cl +} H3PO3}\]
- Reaction with Thionyl chloride(Sulphonyl Chloride)
\[\ce{\underset{\text{Ethanol}}{CH3CH3OH} + SOCl2 ->[Pyridine] \underset{\text{Chloro ethane}}{CH3CH2Cl +} SO2↑ + HCl↑}\]
APPEARS IN
संबंधित प्रश्न
With respect to the position of – Cl in the compound CH3 – CH = CH – CH2 – Cl, it is classified as ______.
Match the compounds given in Column I with suitable items given in Column II.
| Column I (Compound) | Column II (Uses) |
||
| A | Iodoform | 1 | Fire extinguisher |
| B | Carbon tetra chloride | 2 | Insecticide |
| C | CFC | 3 | Antiseptic |
| D | DDT | 4 | Refrigerants |
Which one of the following is most reactive towards nucleophilic substitution reaction?
Give reasons for the polarity of C – X bond in haloalkane.
Compare SN1 and SN2 reaction mechanisms.
In an experiment ethyliodide in ether is allowed to stand over magnesium pieces. Magnesium dissolves and product is formed
- Name the product and write the equation for the reaction.
- Why all the reagents used in the reaction should be dry? Explain.
- How is acetone prepared from the product obtained in the experiment.
Predict the product when Bromo ethane is treated with the following.
KNO2
Starting from CH3MgI, How will you prepare the following?
Ethyl acetate
Starting from CH3MgI, How will you prepare the following?
Iso propyl alcohol
The simplest alkene (A) reacts with HCl to form a compound (B). Compound (B) reacts with ammonia to form compound (C) of molecular formula C2H7N. Compound (C) undergoes carbylamine test. Identify (A),
(B) and (C).
