Advertisements
Advertisements
Question
Mention any three methods of preparation of haloalkanes from alcohols.
Solution
Haloalkanes are prepared by the following methods.
From alcohols: Alcohol can be converted into halo alkenes by reacting it with any one of the following reagents.
- Hydrogen halide
- Phosphorous halides
- Thionyl chloride.
- Reaction with hydrogen halide:
\[\ce{\underset{\text{Ethanol}}{CH3CH2}OH + HCl ->[Anhydrous ZnCl2][\Delta] \underset{\text{Chloroethane}}{CH3CH2Cl + H2 O}}\]
Mixture of con. HCl and anhydrous ZnCl2 is called Lucas Reagent.
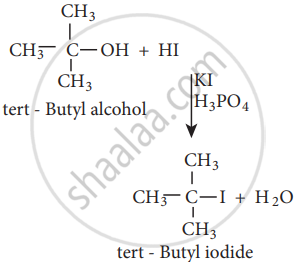
The order of reactivity of halo acids with alcohol is in the order HI > HBr > HCl.
The order of reactivity of alcohols with halo acid is tertiary > secondary > primary.
- Reaction with phosphorous halides:
Alcohols react with PX5 or PX3 to form haloalkanes.
Example:
\[\ce{\underset{\text{Ethane}}{CH3CH2}OH + PCl5 ->\underset{\text{Chloro ethane}}{CH3CH2Cl +}POCl3 + HCl}\]
\[\ce{\underset{\text{Ethanol}}{3CH3CH2OH} + PCl3 -> \underset{\text{Chloro ethane}}{3 CH3CH2Cl +} H3PO3}\]
- Reaction with Thionyl chloride(Sulphonyl Chloride)
\[\ce{\underset{\text{Ethanol}}{CH3CH3OH} + SOCl2 ->[Pyridine] \underset{\text{Chloro ethane}}{CH3CH2Cl +} SO2↑ + HCl↑}\]
APPEARS IN
RELATED QUESTIONS
Benzene reacts with Cl2 in the presence of FeCl3 and in absence of sunlight to form ______.
Which one of the following is most reactive towards nucleophilic substitution reaction?
Give reasons for the polarity of C – X bond in haloalkane.
p – dichlorobenzene has a higher melting point than those of o – and m – dichlorobenzene.
Predict the product when Bromo ethane is treated with the following.
KNO2
Write a short note on Dows Process.
Starting from CH3MgI, How will you prepare the following?
Iso propyl alcohol
Complete the following reaction.
\[\ce{CH3 - CH = CH2 + HBr ->[Peroxide]}\]
Complete the following reaction.
\[\ce{CH3 - CH2 - Br + NaSH ->[alcohol][H2O]}\]
The simplest alkene (A) reacts with HCl to form a compound (B). Compound (B) reacts with ammonia to form compound (C) of molecular formula C2H7N. Compound (C) undergoes carbylamine test. Identify (A),
(B) and (C).
