Advertisements
Advertisements
Question
Give reasons for the polarity of C – X bond in haloalkane.
Solution
- Carbon halogen bond is a polar bond as halogens are more electronegative than carbon. The carbon atom exhibits a partial positive charge (δ+) and halogen atom a partial negative charge (δ–).
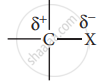
- The C -X bond is formed by the overlap of sp3 orbital of a carbon atom with the half-filled p- orbital of the halogen atom. The atomic size of halogen increases from fluorine to iodine, which increases the C – X bond length. Larger the size, greater is the bond length, and the weaker is the bond formed. The bond strength of C – X decreases from C – F to C – I in CH3X.
APPEARS IN
RELATED QUESTIONS
Of the following compounds, which has the highest boiling point?
Silver propionate when refluxed with Bromine in carbon tetrachloride gives ______.
Compare SN1 and SN2 reaction mechanisms.
p – dichlorobenzene has a higher melting point than those of o – and m – dichlorobenzene.
In an experiment ethyliodide in ether is allowed to stand over magnesium pieces. Magnesium dissolves and product is formed
- Name the product and write the equation for the reaction.
- Why all the reagents used in the reaction should be dry? Explain.
- How is acetone prepared from the product obtained in the experiment.
Predict the product when Bromo ethane is treated with the following.
KNO2
Starting from CH3MgI, How will you prepare the following?
Iso propyl alcohol
Complete the following reaction.
\[\ce{CH3 - CH = CH2 + HBr ->[Peroxide]}\]
Complete the following reaction.
\[\ce{CH3 - CH2 - Br + NaSH ->[alcohol][H2O]}\]
The simplest alkene (A) reacts with HCl to form a compound (B). Compound (B) reacts with ammonia to form compound (C) of molecular formula C2H7N. Compound (C) undergoes carbylamine test. Identify (A),
(B) and (C).
