Advertisements
Advertisements
प्रश्न
Name the chemical compound which is electrolysed in molten state to obtain sodium metal. Which gas is produced in this process?
उत्तर
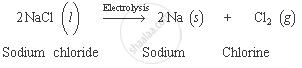
Sodium chloride is electrolysed in molten state to obtain sodium metal. Chlorine gas is evolved during this process.
The equation for this decomposition is:
APPEARS IN
संबंधित प्रश्न
In the extraction of aluminium: Name the process of concentration of bauxite.
In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
Define the term mineral.
Name two metals which are found in nature mainly in the free state (as metallic elements).
Name three other metals which are extracted in a manner similar to sodium.
Some metallic oxides can be reduced by hydrogen, carbon and carbon monoxide and some cannot. Explain.
Name the following:
Name two metals always find in combined state.
Write the molecular formulae of the following compound.
Cryolite
2 mL each of concentrated HCl, HNO3 and a mixture of concentrated HCl and concentrated HNO3 in the ratio of 3 : 1 were taken in test tubes labelled as A, B and C. A small piece of metal was put in each test tube. No change occurred in test tubes A and B but the metal got dissolved in test tube C respectively. The metal could be
Iqbal treated a lustrous, divalent element M with sodium hydroxide. He observed the formation of bubbles in reaction mixture. He made the same observations when this element was treated with hydrochloric acid. Suggest how can he identify the produced gas. Write chemical equations for both the reactions.
