Advertisements
Advertisements
Question
Name the chemical compound which is electrolysed in molten state to obtain sodium metal. Which gas is produced in this process?
Solution
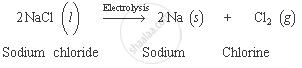
Sodium chloride is electrolysed in molten state to obtain sodium metal. Chlorine gas is evolved during this process.
The equation for this decomposition is:
APPEARS IN
RELATED QUESTIONS
Why do gold ornaments look new even after several years of use?
Which one of the methods given in column I is applied for the extraction of each of the metals given in column II:
| Column I | Column II |
| Electrolytic reduction | Aluminium |
| Reduction with Carbon | Zinc |
| Reduction with Aluminium | Sodium |
| Iron | |
| Manganese | |
| Tin |
Name the electrode at which aluminium metal is produced.
Which of the following metals are extracted by the electrolysis of their molten chlorides?
(a) Na and Hg
(b) Hg and Mg
(c) Na and Mg
(d) Cu and Fe
What is meant by concentration of ores?
Complete the incomplete statement with missing word:
Metals are malleable while non-metals are ______.
Name the following :
The Process of heating an ore to a high temperature in the presence of air.
Find the odd one out and give its explanation.
The electrolysis of alumina involves the use of fluorspar and cryolite to increase the melting point.
- Given below are the steps for extraction of copper from its ore. Write the reaction involved.
- Roasting of copper (I) sulphide
- Reduction of copper (I) oxide with copper (I) sulphide.
- Electrolytic refining
- Draw a neat and well labelled diagram for electrolytic refining of copper
