Advertisements
Advertisements
Question
- Given below are the steps for extraction of copper from its ore. Write the reaction involved.
- Roasting of copper (I) sulphide
- Reduction of copper (I) oxide with copper (I) sulphide.
- Electrolytic refining
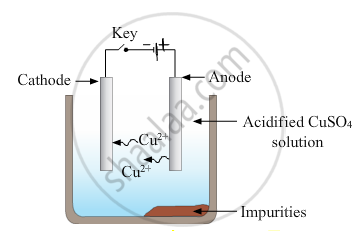
- Draw a neat and well labelled diagram for electrolytic refining of copper
Solution
- `2"Cu"_2"S" + 3"O"_2 overset("Heat")(->)2"Cu"_2"O" + 2"SO"_2`
- `2"Cu"_2"O" + "Cu"_2"S" overset("Heat")(->) 6"Cu" + "SO"_2 `
- Electrode reactions :
Reaction at anode - `"Cu" -> "Cu"^(2+) + 2^("e"-)`
Reaction at cathode - `"Cu"^(2+) + 2"e"^- -> "Cu"`
APPEARS IN
RELATED QUESTIONS
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. This oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context, answer the following:
1) What kind of combination exists between M and O?
2)How many electrons are there in the outermost shell of M?
3) Name the group to which M belongs.
4) State the reaction taking place at the cathode.
5) Name the product at the anode.
How is zinc extracted from its carbonate or (calamine)? Explain with equations.
What iron compound is present in haematite ore? Also write its chemical formula.
What is the difference between a mineral and an ore?
An ore of manganese metal is:
(a) bauxite
(b) haematite
(c) cuprite
(d) pyrolusite
Define the following term.
Mineral
Give the chemical formula of :
Bauxite
Define the term : Flux
Explain the following:
Galvanization protects iron from rusting.
The highly reactive metals like Sodium, Potassium, Magnesium, etc. are extracted by the ____________.
