Advertisements
Advertisements
प्रश्न
- Given below are the steps for extraction of copper from its ore. Write the reaction involved.
- Roasting of copper (I) sulphide
- Reduction of copper (I) oxide with copper (I) sulphide.
- Electrolytic refining
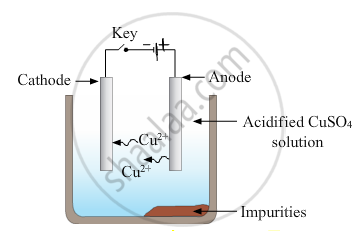
- Draw a neat and well labelled diagram for electrolytic refining of copper
उत्तर
- `2"Cu"_2"S" + 3"O"_2 overset("Heat")(->)2"Cu"_2"O" + 2"SO"_2`
- `2"Cu"_2"O" + "Cu"_2"S" overset("Heat")(->) 6"Cu" + "SO"_2 `
- Electrode reactions :
Reaction at anode - `"Cu" -> "Cu"^(2+) + 2^("e"-)`
Reaction at cathode - `"Cu"^(2+) + 2"e"^- -> "Cu"`
APPEARS IN
संबंधित प्रश्न
A zinc ore on heating in air forms sulphur dioxide. Describe briefly any two stages involved in the conversion of this concentrated ore into zinc metal.
Name two ores of zinc. Write the names of the chemical compounds present in them and give their chemical formulae.
Which of the following is an ore of mercury metal?
(a) rock salt
(b) cinnabar
(c) calamine
(d) haematite
State three objectives achieved during the roasting of ores.
Give reasons, why aluminum is used in:
In making ships
Complete the incomplete statement with missing word:
Metals are malleable while non-metals are ______.
Explain the characteristics of ionic compounds.
2 mL each of concentrated HCl, HNO3 and a mixture of concentrated HCl and concentrated HNO3 in the ratio of 3 : 1 were taken in test tubes labelled as A, B and C. A small piece of metal was put in each test tube. No change occurred in test tubes A and B but the metal got dissolved in test tube C respectively. The metal could be
An alloy is
Iqbal treated a lustrous, divalent element M with sodium hydroxide. He observed the formation of bubbles in reaction mixture. He made the same observations when this element was treated with hydrochloric acid. Suggest how can he identify the produced gas. Write chemical equations for both the reactions.
