Advertisements
Advertisements
प्रश्न
- Given below are the steps for extraction of copper from its ore. Write the reaction involved.
- Roasting of copper (I) sulphide
- Reduction of copper (I) oxide with copper (I) sulphide.
- Electrolytic refining
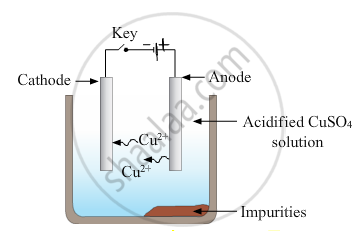
- Draw a neat and well labelled diagram for electrolytic refining of copper
उत्तर
- `2"Cu"_2"S" + 3"O"_2 overset("Heat")(->)2"Cu"_2"O" + 2"SO"_2`
- `2"Cu"_2"O" + "Cu"_2"S" overset("Heat")(->) 6"Cu" + "SO"_2 `
- Electrode reactions :
Reaction at anode - `"Cu" -> "Cu"^(2+) + 2^("e"-)`
Reaction at cathode - `"Cu"^(2+) + 2"e"^- -> "Cu"`
APPEARS IN
संबंधित प्रश्न
Silver metal does not combine easily with oxygen but silver jewellery tarnishes after some time. How?
State three objectives achieved during the roasting of ores.
What is galvanized iron.
How many valence electrons are present in non- metals?
Which one of the following is not true of metal :
Metals form non-polar covalent compounds
Define roasting.
Complete flow chart given below.
Answer the questions on the following passage.
The minerals from which the metal can be separated economically are called ores. Ores contain many types of impurities such as soil, sand and rocky substances along with metal compounds. These impurities are called gangue.
Metals can be extracted from their ores by means of various methods of separation. The process of extraction of metal in a pure state from the ores is also a part of metallurgy.
Ores are taken out from the mines and the gangue is usually separated from the ore at the site itself by various methods. Then the ores are carried out to the place where metals are produced. Here metals are extracted in pure form. Then metals are further purified by different methods of purification. This entire process is called metallurgy. Most metals being reactive do not occur in nature in the free state but are found in combined state as their salts such as oxides, carbonates, sulphides, and nitrates. however, the most unreactive metals that are not affected by air, water and other natural factors like silver, gold, platinum, generally occur in a free state. The compounds of metals that occur in nature along with the impurities are called minerals.
- What are ores?
- Which processes are involved in the branch of metallurgy? What is metallurgy?
- Which metals are found in a free state?
- In what forms are metals found in combined state?
- What is gangue?
Electrical wires have a coating of an insulting material. The material, generally used is
On the basis of reactivity metals are grouped into three categories:
- Metals of low reactivity
- Metals of medium reactivity
- Metals of high reactivity
Therefore metals are extracted in pure form from their ores on the basis of their chemical properties.
Metals of high reactivity are extracted from their ores by electrolysis of the molten ore.
Metals of low reactivity are extracted from their sulphide ores, which are converted into their oxides. The oxides of these metals are reduced to metals by simple heating.
(a) Name the process of reduction used for a metal that gives vigorous reaction with air and water both.
(b) Carbon cannot be used as a reducing agent to obtain aluminium from its oxide? Why?
(c) Describe briefly the method to obtain mercury from cinnabar. Write the chemical equation for the reactions involved in the process.
OR
(c) Differentiate between roasting and calcination giving chemical equation for each.
