Advertisements
Advertisements
प्रश्न
Name the Following:
Product obtained by the catalytic oxidation of ammonia.
उत्तर
Product obtained by catalytic oxidation of ammonia are NO, water and heat.
APPEARS IN
संबंधित प्रश्न
State one relevant observation for the following:
When crystals of copper nitrate are heated in a test tube.
Write balanced equations for action of warm water on magnesium nitride
What is fixation of Nitrogen?
Nitric acid cannot be concentrated beyond 68% by the distillation of a dilute solution of \[\ce{HNO3}\]. State the reason.
Identify the substance underlined:
The dilute acid which is an oxidizing agent.
Write equation to show the reaction between the following:
Copper oxide and dilute nitric acid.
Write balanced equation to show the reaction between the sulphur and hot concentrated nitric acid.
Write a balanced equation for following :
Action of cold and dilute nitric acid on copper
Write sqauential observation for effect of heat on Copper nitrate.
The diagram given below is a representation of the Industrial preparation of Nitric acid by Ostwald’s process. With respect to the process answer the following questions:
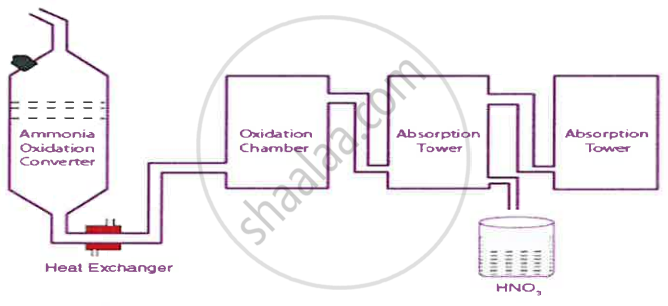
- Write the temperature and the catalyst required during the catalytic oxidation of ammonia.
- Give balanced chemical equation for the reaction occurring duringthe conversion of nitrogen dioxide to nitric acid.
