Advertisements
Advertisements
प्रश्न
What is fixation of Nitrogen?
उत्तर
The conversion of free atmospheric nitrogen into useful nitrogenous compounds in the soil is known as the fixation of atmospheric nitrogen.
APPEARS IN
संबंधित प्रश्न
Name the gas that is produced in the given case:
The action of cold and dilute nitric acid on copper
What is aqua fortis?
During thunderstorm, rain water contains nitric acid. Explain with reactions.
Give reasons for the following:
In the laboratory preparation of nitric acid, the mixture of concentrated sulphuric acid and sodium nitrate should not be heated very strongly above 200°C.
Name the Following:
Product obtained by the catalytic oxidation of ammonia.
Write the balanced equation for the following:
Action of concentrated nitric acid on copper.
How would you separate gold from a mixture of gold and copper filling?
Write down the word equation or balanced equation for the action of concentrated nitric acid on copper.
Fill in the blank using the appropriate words given below:
Hot, concentrated nitric add reacts with sulphur to form ______.
The diagram given below is a representation of the Industrial preparation of Nitric acid by Ostwald’s process. With respect to the process answer the following questions:
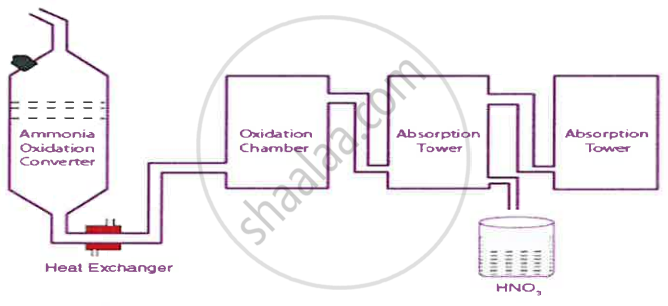
- Write the temperature and the catalyst required during the catalytic oxidation of ammonia.
- Give balanced chemical equation for the reaction occurring duringthe conversion of nitrogen dioxide to nitric acid.
