Advertisements
Advertisements
प्रश्न
Name the gas evolved when zinc granules are treated/heated with:
hydrochloric acid solution
उत्तर
When zinc granules are treated with hydrochloric acid, zinc chloride is formed and hydrogen gas is liberated.
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
APPEARS IN
संबंधित प्रश्न
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with zinc granules.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with magnesium ribbon.
Fill in the blank in the following sentences:
Substances do not show their acidic properties without.......................... .
What happens when an acid reacts with a metal hydrogencarbonate? Write equation of the reaction which takes place.
What ions are present in the solutions of following substances? (write the symbols only)
Hydrochloric acid
Define an acid and a base. Give two examples of each.
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Write the chemical equation of the reaction of metal X with (i) hydrochloric acid, and (ii) sodium hydroxide solution.
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
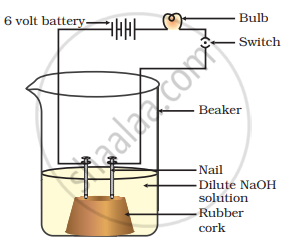
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Are all acids corrosive in nature? Name a few acids which are non-corrosive and may be part of our food.
Copper or brass cooking vessels are coated with tin metal. Why?
