Advertisements
Advertisements
प्रश्न
Name the gas evolved when zinc granules are treated/heated with:
sodium hydroxide solution
What happens when zinc granules are heated with sodium hydroxide solution? Write equation of the reaction which takes place.
उत्तर १
Zinc reacts with sodium hydroxide, giving sodium zincate and hydrogen gas.
Zn(s) + 2NaOH(aq)→ Na2ZnO2(aq) + H2(g)
उत्तर २
When zinc granules are heated with sodium hydroxide (base), sodium zincate salt is formed with the liberation of hydrogen gas.
Zn (s) + 2 NaOH(aq)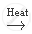 Na2 Zn O2 (aq) + H2 (g)
Na2 Zn O2 (aq) + H2 (g)
संबंधित प्रश्न
Give the names and formulae of two strong acids and two weak acids.
Name one natural source of the following acids:
Citric acid
Name one animal and one plant whose stings contain formic acid (or methanoic acid).
What colour does phenolphthalein indicator turn when added to an alkali (such as sodium hydroxide)?
What happens during a bee sting? What is its remedy?
Name a sodium compound which is a constituent of many dry soap powders.
What type of ions is formed:
when a base (or alkali) is dissolved in water?
Define a base.
In acidic solutions blue litmus changes into ______ colour.
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
(i) Temperature of the solution decreases.
(ii) Temperature of the solution increases.
(iii) Temperature of the solution remains the same.
(iv) Salt formation takes place.
