Advertisements
Advertisements
प्रश्न
Name the gas evolved when zinc granules are treated/heated with:
sodium hydroxide solution
What happens when zinc granules are heated with sodium hydroxide solution? Write equation of the reaction which takes place.
उत्तर १
Zinc reacts with sodium hydroxide, giving sodium zincate and hydrogen gas.
Zn(s) + 2NaOH(aq)→ Na2ZnO2(aq) + H2(g)
उत्तर २
When zinc granules are heated with sodium hydroxide (base), sodium zincate salt is formed with the liberation of hydrogen gas.
Zn (s) + 2 NaOH(aq)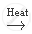 Na2 Zn O2 (aq) + H2 (g)
Na2 Zn O2 (aq) + H2 (g)
संबंधित प्रश्न
Which gas is usually liberated when an acid reacts with a metal?
Explian the following:
Dil. HCl acid is stronger than highly concentrated acetic acid.
what effect does the concentration of [H3O+] ion have on solution?
Write balanced equation to satisfy the following statement:
\[\ce{Acid + Sulphite or bisulphite -> Salt + water + sulphur dioxide}\]
Table salt is acidic.
Give the name and formula of two : Non-volatile acids
Explain, all alkalis are bases but all bases are not alkalis.
Acids are bitter in taste.
Acid - Define.
Complete the following equation.
Zn + ______ → Na2ZnO2 + H2 ↑
