Advertisements
Advertisements
प्रश्न
Name the processes from which chlorine is obtained as a by-product. What will happen if an aqueous solution of NaCl is subjected to electrolysis?
उत्तर
In the electrolysis of molten NaCl, Cl2 is obtained at the anode as a by product.
`NaCl_(("melt")) -> Na_( ("melt"))^+ + Cl_( ("melt"))^(-)`
At cathode: `Na_( ("melt"))^+ e^(-) -> Na_((s))`
At anode : `Cl_( ("melt"))^+ -> Cl_((g)) + e^(-)`
`2Cl_((g)) -> Cl_(2(g))`
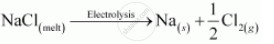
The overall reaction is as follows:
If an aqueous solution of NaCl is electrolyzed, Cl2 will be obtained at the anode but at the cathode, H2will be obtained (instead of Na). This is because the standard reduction potential of Na (E°= − 2.71 V) is more negative than that of H2O (E° = − 0.83 V). Hence, H2O will get preference to get reduced at the cathode and as a result, H2 is evolved.
`NaCl_( (aq)) -> Na_( (aq))^+ + Cl_((aq))^-`
At cathod: `2H_2O_((l)) + 2e^(-) -> H_(2(g)) + 2OH_((aq))^-`
At anode : `Cl_(("melt"))^(-) -> Cl_((g)) + e^(-)`
`2Cl_((g)) -> Cl_(2(g))`
APPEARS IN
संबंधित प्रश्न
What is calcination?
Explain calcination with reactions.
What is the process in which concentrated ore is reduced to the corresponding metal by heating at high temperature with a reducing agent?
(a) Polling
(b) Pyrometallurgy
(c) Hydrometallurgy
(d) Calcination
Reduction of metal oxide to metal becomes easier if the metal obtained is in liquid state. Why?
What is the role of zinc metal in the extraction of silver ?
MnO2 and Ca3(PO4)2 present in iron ore get reduced to Mn and P in the zone of _______.
(A) combustion
(B) reduction
(C) fusion
(D) slag formation
Which one of the following oxidation state of Manganese is unstable?
(a) + 2
(b) + 4
(c) + 5
(d) + 7
Complete the given chemical equations
`F_2 + 2Cl^(-)`
What is the action of carbon on the following metal oxides:
Fe2O3 in the blast furnace
What is the action of carbon on the following metal oxides :
ZnO in the vertical retort furnace
An element X (molar mass = 60 g mol−1) has a density of 6.23 g cm−3. Identify the type of cubic unit cell, if the edge length of the unit cell is 4 ✕ 10−8 cm.
Which metal is also found in sea beds?
In the extraction of chlorine by electrolysis of brine ______.
Brine is electrolysed by using inert electrodes. The reaction at anode is ______.
At temperatures above 1073 K coke can be used to reduce \[\ce{FeO}\] to \[\ce{Fe}\]. How can you justify this reduction with Ellingham diagram?
Why are sulphide ores converted to oxide before reduction?
