Advertisements
Advertisements
प्रश्न
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
hydrogen
उत्तर
In the presence of nickel, platinum, or palladium, ethyne changes to ethene and then to ethane.
\[\ce{\underset{Ethyne}{CH ≡ CH} + H2 ->[Ni][300^\circ C] \underset{Ethene}{CH2 = CH2} + H2 ->[Ni][300^\circ C] \underset{Ethane}{CH3 - CH3}}\]
APPEARS IN
संबंधित प्रश्न
The compound formed where two alkyl groups are linked by 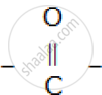 group.
group.
Write the structure of the following compound:
2 – methylpropane
Write the structure of the following compound:
3-hexene
What are the sources for alkynes?
Give a chemical test to distinguish between ethene (ethylene) and ethyne (acetylene).
Name the products formed and write an equation when ethyne is added to the hydrogen in an
inert solvent ?
Name the hydrocarbon which is a planar molecule.
Name the hydrocarbon which is a linear molecule.
Give the chemical equation for the preparation of ethyne from 1, 2-dibromoethane.
Alkynes undergo ______ reactions.
