Advertisements
Advertisements
Question
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
hydrogen
Solution
In the presence of nickel, platinum, or palladium, ethyne changes to ethene and then to ethane.
\[\ce{\underset{Ethyne}{CH ≡ CH} + H2 ->[Ni][300^\circ C] \underset{Ethene}{CH2 = CH2} + H2 ->[Ni][300^\circ C] \underset{Ethane}{CH3 - CH3}}\]
APPEARS IN
RELATED QUESTIONS
The compound formed where two alkyl groups are linked by 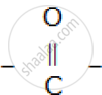 group.
group.
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulphide, ethyne]
This gas is used for welding purposes.
State the conditions required for the given reaction to take place:
Catalytic hydrogenation of ethyne
Write the structure of the following compound:
Prop-1-ene
Write the structure of the following compound:
2 – methylpropane
Write the structure of the following compound:
3-hexene
Name the products formed and write an equation when ethyne is added to the bromine in an
inert solvent ?
Name a triple bond hydrocarbon with two carbon atoms.
State how the following conversion can be carried out:
Ethyl chloride to Ethyl alcohol
Give a balanced equation for the following conversion.
An alkyne to an alkene.
