Advertisements
Advertisements
प्रश्न
Observe the figure and name and explain in brief the following method:

उत्तर
The method shown in the figure is froth floatation method.
- The froth floatation process is based on two opposite properties of the particles, hydrophilic and hydrophobic.
- The metal sulphide particles are hydrophobic. Due to this property, they get wetted mainly with oil. On the other hand, gangue particles are hydrophilic and get wetted by water.
- In this method, the finely divided metal ore is added in a big tank containing a large amount of water. To this, certain vegetable oil (pine oil, eucalyptus oil, etc.) is added is added for the formation of froth. A rotating agitator at the centre of the floatation tank agitates the mixture and draws air into it to form bubbles. As a result, foam is formed which carries metal sulphide particles. The foam rises to the surface of water and floats. Therefore, this method is called the froth floatation process.
- This method is used to separate metal sulphide ores such as zinc blende (ZnS) and copper pyrite (CuFeS2).
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Why are alkali metals kept in kerosene oil?
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: carbonates
Name:
(a) the processes involved in
concentration :-
Explain the following terms:
(a) flux
With a labelled diagram, explain the electro-refining of a particular metal.
`Ag2O` ................................
................................
`HgS + O_2`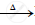 ..................
..................
Where are the cathode and anode in the electrolytic cell? Name the material used for these?
Write the molecular formulae of the following compound.
Copper pyrite
Distinguish between:
Froth floatation - Leaching
Identify the following method of concentration of ores and explain briefly.

Observe the figure and name and explain in brief the following method:

Bassemerisation is used in the extraction of ______.
Limestone is used as a flux in the extraction of ______.
Which of following metals occurs in native state?
In leaching of alumina from bauxite by Bayer's process, then ore is treated with ______.
After partial roasting, the sulphide of copper is reduced by ______.
Calcination is used in metallurgy for the removal of?
