Advertisements
Advertisements
प्रश्न
With a labelled diagram, explain the electro-refining of a particular metal.
आकृति
स्पष्ट कीजिए
उत्तर
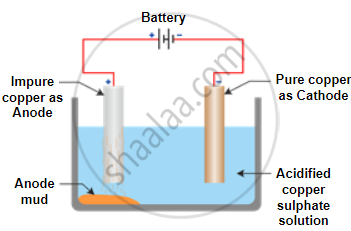
The process of electrolytic refining copper:
- In this electrolytic refining, the electrolyte is a solution of copper sulphate.
- In this process, the anode is impure copper, whereas the cathode is a thin strip of pure copper.
- On passing the current through the electrolyte, the pure copper from the anode dissolves into the electrolyte.
- An equivalent amount of pure copper from the electrolyte is deposited at the cathode.
- The insoluble impurities that settle down at the bottom of the anode (the positively charged electrode) are known as anode mud.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: nitrates
`Ag2O` ................................
................................
`2Cu_2O +Cu_2S` ......................
......................
How is it removed?
Name the following:
Name an allotrope of a non-metal that allows electricity to pass through it.
The chemical formula of zinc blend is _______.
Write the molecular formulae of the following compound.
Ferrous tungstate
Observe the figure and name and explain in brief the following method:

Zirconium is refined by ____________.
Bassemerisation is used in the extraction of ______.
