Advertisements
Advertisements
प्रश्न
With a labelled diagram, explain the electro-refining of a particular metal.
आकृती
स्पष्ट करा
उत्तर
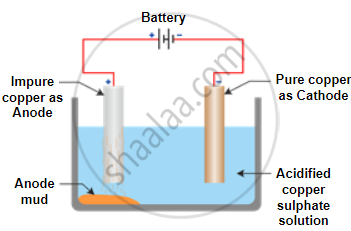
The process of electrolytic refining copper:
- In this electrolytic refining, the electrolyte is a solution of copper sulphate.
- In this process, the anode is impure copper, whereas the cathode is a thin strip of pure copper.
- On passing the current through the electrolyte, the pure copper from the anode dissolves into the electrolyte.
- An equivalent amount of pure copper from the electrolyte is deposited at the cathode.
- The insoluble impurities that settle down at the bottom of the anode (the positively charged electrode) are known as anode mud.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
Explain the following terms:
smelting
On which factors does the purification of metals depend?
`ZnCO_3`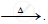 ..............................
..............................
`2AgNO_3` ...........................
...........................
`HgS + O_2`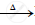 ..................
..................
How is it removed?
Write scientific reason.
Magnetic separation method is used to separate the magnetic ingredients in the ores.
Explain concept with example/explain with the help of a balanced equation.
Gangue
Explain the froth floatation method with a neat labelled diagram.
A process of extracting metals from aqueous solutions of their salts using suitable reducing agents is called ______
