Advertisements
Advertisements
प्रश्न
Explain the froth floatation method with a neat labelled diagram.
उत्तर
- The froth floatation process is based on two opposite properties of the particles, hydrophilic and hydrophobic.
- The metal sulphide particles are hydrophobic. Due to this property, they get wetted mainly with oil. On the other hand, gangue particles are hydrophilic and get wetted by water.
- In this method, the finely divided metal ore is added in a big tank containing large amount of water. To this, certain vegetable oil (pine oil, eucalyptus oil, etc.) is added for the formation of froth.
- In the tank, compressed air is circulated through the water. Bubbles are formed due to this blown air. A rotating agitator at the centre of the floatation tank agitates the mixture and draws air into it to form bubbles. As a result, foam is formed which carries metal sulphide particles. The foam rises to the surface of water and floats. Therefore, this method is called froth floatation process.
- This method is used to separate metal sulphide ores such as zinc blende (ZnS) and copper pyrite (CuFeS2).

Froth floatation method
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Carbon
Name the processes involved in refining of ores.
Explain the following terms:
smelting
`Ag2O` ................................
................................
`2NaNO_3` ......................
......................
`2AgNO_3` ...........................
...........................
`HgS + O_2`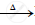 ..................
..................
Give the ionization reactions of electrolyte used in Hall’s process. write the reaction at the cathode and the anode. Why the anode has to be replaced in this process?
what does it form with silica present in the ore?
Which metal is used for:
making pipes, buckets, water tanks,
Which metal is used for:
making face creams
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
Why does iron or zinc not occur free in nature?
Distinguish between:
Froth floatation - Leaching
Explain concept with example/explain with the help of a balanced equation.
Gangue
Explain concept with example/explain with the help of a balanced equation.
Minerals
Observe the figure and name and explain in brief the following method:

Calculate the difference between heat of combustion of carbon monoxide gas at constant pressure and at constant volume at 27°C? (R = 2 cal K-1 mol-1).
Bassemerisation is used in the extraction of ______.
Name the process that is employed to refine aluminium.
