Advertisements
Advertisements
प्रश्न
Explain concept with example/explain with the help of a balanced equation.
Minerals
उत्तर
- Most metals being reactive do not occur in nature in the free state but are found in a combined state as their salts such as oxides, carbonates, sulphides, and nitrates.
- However, some unreactive metals like silver, gold, platinum, generally occur in a free state.
- The compounds of metals that occur in nature along with the impurities are called minerals.
- For example, bauxite is a mineral of aluminium.
APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: nitrates
Define the term ‘metallurgy’. State the processes involved in metallurgy ?
Name:
(b) two metallic oxides which cannot be reduced by carbon, carbon monoxide or hydrogen
On which factors does the purification of metals depend?
With a labelled diagram, explain the electro-refining of a particular metal.
`Ag2O` ................................
................................
`2Pb(NO3)_2`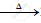 .........................
.........................
Give the ionization reactions of electrolyte used in Hall’s process. write the reaction at the cathode and the anode. Why the anode has to be replaced in this process?
Why is flux used in the blast furnace?
what does it form with silica present in the ore?
Distinguish between electrolytic methods of reduction and refining.
Which metal is used for:
making pipes, buckets, water tanks,
Which metal is used for:
making face creams
In Wilfley table method to separate particles of gangue _______ method is used.
Cassiterite is a copper ore.
Write scientific reason.
Magnetic separation method is used to separate the magnetic ingredients in the ores.
Observe the figure and name and explain in brief the following method:

State the reason for the addition of caustic alkali to bauxite ore during the purification of bauxite.
In leaching of alumina from bauxite by Bayer's process, then ore is treated with ______.
