Advertisements
Advertisements
प्रश्न
Explain concept with example/explain with the help of a balanced equation.
Minerals
उत्तर
- Most metals being reactive do not occur in nature in the free state but are found in a combined state as their salts such as oxides, carbonates, sulphides, and nitrates.
- However, some unreactive metals like silver, gold, platinum, generally occur in a free state.
- The compounds of metals that occur in nature along with the impurities are called minerals.
- For example, bauxite is a mineral of aluminium.
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Carbon
brown powder deposit on iron?
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: nitrates
Explain the following terms:
(a) flux
Explain the following terms:
(c) slag
An ore on being heated in air forms sulphurous anhydride. Write the process used for the concentration of this ore.
Name the methods used for purification?
Why is flux used in the blast furnace?
Explain the term of Metallurgy .
Complete the following flow chart and answer the questions below:
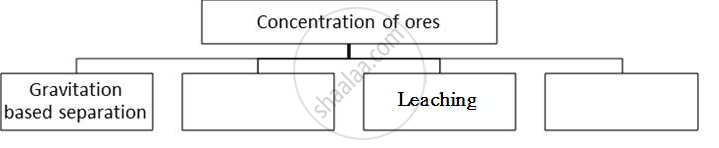
i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
Cassiterite is a copper ore.
Complete flow chart given below.
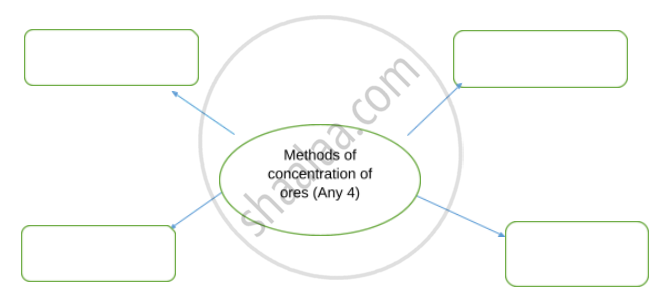
Explain the hydraulic separation method with a neat labelled diagram.
Observe the figure and name and explain in brief the following method:

Observe the figure and name and explain in brief the following method:

Explain the smelting process.
Calcination is used in metallurgy for the removal of?
Name the process that is employed to refine aluminium.
