Advertisements
Advertisements
प्रश्न
In Wilfley table method to separate particles of gangue _______ method is used.
पर्याय
Magnetic
Froth floatation
Leaching
gravitation
उत्तर
In Wilfley table method to separate particles of gangue gravitation method is used.
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Hydrogen
Give the importance of the following for living beings:
Carbon
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: nitrates
Explain the following terms:
smelting
what does it form with silica present in the ore?
Which metal is used for:
making face creams
Name a metal which forms a liquid alloy at ordinary temperature.
Complete the following flow chart and answer the questions below:
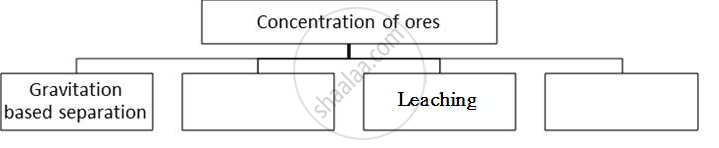
i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
The chemical formula of zinc blend is _______.
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
Explain concept with example/explain with the help of a balanced equation.
Minerals
Explain the hydraulic separation method with a neat labelled diagram.
Which of the following methods is used to separate wolframite and stannic oxide present in cassiterite?
Calculate the difference between heat of combustion of carbon monoxide gas at constant pressure and at constant volume at 27°C? (R = 2 cal K-1 mol-1).
Limestone is used as a flux in the extraction of ______.
Which of following metals occurs in native state?
A process of extracting metals from aqueous solutions of their salts using suitable reducing agents is called ______
Explain the following term:
Metallurgy
