Advertisements
Advertisements
प्रश्न
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
उत्तर
| Group A | Group B |
| 1. ZnS | d) Zinc blend |
| 2. HgS | c) Cinnabar |
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Give the importance of the following for living beings:
Carbon
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
Define the term ‘metallurgy’. State the processes involved in metallurgy ?
Name:
(b) two metallic oxides which cannot be reduced by carbon, carbon monoxide or hydrogen
Explain the following terms:
(a) flux
`2Pb(NO3)_2`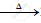 .........................
.........................
`HgS + O_2`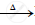 ..................
..................
state the reactions at the two electrodes ?
Why is flux used in the blast furnace?
How is it removed?
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
Cassiterite is a copper ore.
Write the molecular formulae of the following compound.
Ferrous tungstate
Distinguish between:
Froth floatation - Leaching
Explain the froth floatation method with a neat labelled diagram.
The chemical name of rust is ______.
In leaching of alumina from bauxite by Bayer's process, then ore is treated with ______.
Calcination is used in metallurgy for the removal of?
Name the process that is employed to refine aluminium.
