Advertisements
Advertisements
Question
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
Solution
| Group A | Group B |
| 1. ZnS | d) Zinc blend |
| 2. HgS | c) Cinnabar |
APPEARS IN
RELATED QUESTIONS
Give the importance of the following for living beings:
Hydrogen
Give the importance of the following for living beings:
Carbon
A student has been collecting silver coins and copper coins. One day she observed a black
coating on silver coins and a green coating on copper coins. Which chemical phenomenon is
responsible for these coatings? Write the names of black and green coatings.
Name:
(a) the processes involved in
concentration :-
Explain the following terms:
(c) slag
`Ag2O` ................................
................................
`HgS + O_2`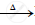 ..................
..................
Which metal is used for:
making face creams
State two properties of brass that render it more useful for some purpose than its components.
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
Explain the term of Metallurgy .
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
Why does iron or zinc not occur free in nature?
In Wilfley table method to separate particles of gangue _______ method is used.
The chemical formula of zinc blend is _______.
Explain the hydraulic separation method with a neat labelled diagram.
Explain the froth floatation method with a neat labelled diagram.
Calculate the difference between heat of combustion of carbon monoxide gas at constant pressure and at constant volume at 27°C? (R = 2 cal K-1 mol-1).
