Advertisements
Advertisements
प्रश्न
Out of [CoF6]3- and [Co(en)3]3+, which one complex is
(i) paramagnetic
(ii) more stable
(iii) inner orbital complex and
(iv) high spin complex
(Atomic no. of Co = 27)
उत्तर
Co (Z=27)=1s2 2s2 2p6 3s2 3p6 4s2 3d7
Co+3 =1s2 2s2 2p6 3s2 3p6 3d7
F- is weak field ligand:
[CoF6]3-
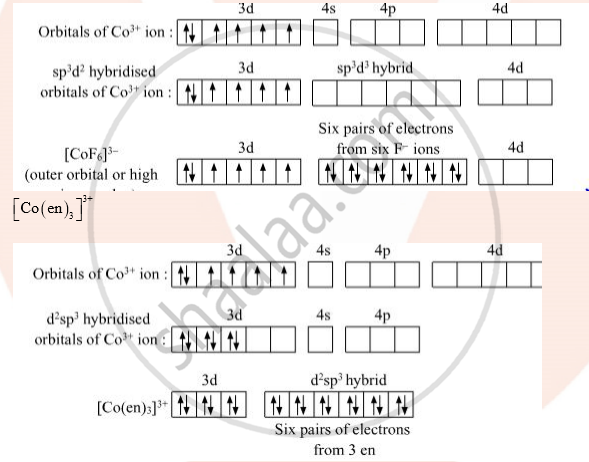
(i) [CoF6]3- is paramagnetic due to the presence of four unpaired electrons
(ii) [Co(en)3]3+ is more stable due to chelation
(iii) [Co(en)3]3+ forms inner orbital complex (d2sp3)
(iv) [CoF6]3+ Forms high spin complex (sp3d2)
APPEARS IN
संबंधित प्रश्न
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Write the type of magnetism observed when the magnetic moments are aligned in parallel and anti-parallel directions in unequal numbers.
Explain the following with suitable examples: Paramagnetism
Give reasons:Ferrimagnetic substances show better magnetism than antiferromagnetic substances.
What happens to the domains in a ferromagnetic material in the presence of external magnetic field?
Substances which are weakly repelled in magnetic field are known as ____________.
Which one of the following would feel attraction when placed in magnetic field: Co2+, Ag+, Ti4+, Zn2+
Among the following ions, which one has the highest paramagnetism?
Which of the following is not paramagnetic?
The metal complex ion that is paramagnetic is ______.
(Atomic number of Fe = 26, Cu = 29, Co = 27 and Ni = 28)
