Advertisements
Advertisements
Question
Out of [CoF6]3- and [Co(en)3]3+, which one complex is
(i) paramagnetic
(ii) more stable
(iii) inner orbital complex and
(iv) high spin complex
(Atomic no. of Co = 27)
Solution
Co (Z=27)=1s2 2s2 2p6 3s2 3p6 4s2 3d7
Co+3 =1s2 2s2 2p6 3s2 3p6 3d7
F- is weak field ligand:
[CoF6]3-
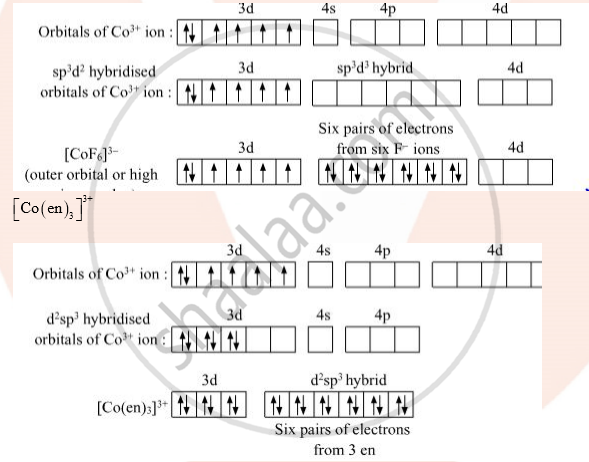
(i) [CoF6]3- is paramagnetic due to the presence of four unpaired electrons
(ii) [Co(en)3]3+ is more stable due to chelation
(iii) [Co(en)3]3+ forms inner orbital complex (d2sp3)
(iv) [CoF6]3+ Forms high spin complex (sp3d2)
APPEARS IN
RELATED QUESTIONS
Write the type of magnetism observed when the magnetic moments are oppositely aligned and cancel out each other.
Write the type of magnetism observed when the magnetic moments are aligned in parallel and anti-parallel directions in unequal numbers.
What type of substances would make better permanent magnets, ferromagnetic or ferrimagnetic. Justify your answer.
Give reasons:Ferrimagnetic substances show better magnetism than antiferromagnetic substances.
Pyrolusite ore is _______.
Compare the properties of soft and hard ferromagnetic materials.
When heated to high temperature, ferromagnetic substance changes to ____________.
The value of magnetic moment is zero in the case of antiferromagnetic substances because the domains:
(i) get oriented in the direction of the applied magnetic field.
(ii) get oriented opposite to the direction of the applied magnetic field.
(iii) are oppositely oriented with respect to each other without the application of magnetic field.
(iv) cancel out each other’s magnetic moment.
Among the following ions, which one has the highest paramagnetism?
The metal complex ion that is paramagnetic is ______.
(Atomic number of Fe = 26, Cu = 29, Co = 27 and Ni = 28)
