Advertisements
Advertisements
प्रश्न
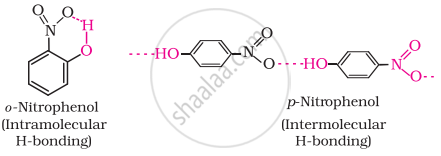
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
उत्तर
The ortho and para isomers can be separated by steam distillation. o-Nitrophenol is steam volatile due to intramolecular hydrogen bonding while p-nitrophenol is less volatile due to intermolecular hydrogen bonding which causes the association of molecules.
APPEARS IN
संबंधित प्रश्न
Write the equation involved in the following reaction:
Kolbe’s reaction
Give the equation of the following reaction:
Bromine in CS2 with phenol.
On distilling phenol with Zn dust, one gets:
In the reaction of phenol with CHCl3 and aqueous NaOH at 343 K, the electrophile attacking the ring is:
When Phenol is distilled with zinc dust, it gives:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:

Which of the following species can act as the strongest base?
Which of the following is not aromatic?
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
