Advertisements
Advertisements
Question
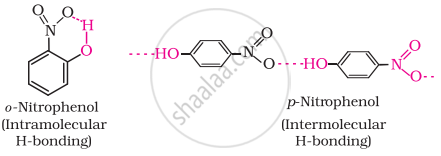
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Solution
The ortho and para isomers can be separated by steam distillation. o-Nitrophenol is steam volatile due to intramolecular hydrogen bonding while p-nitrophenol is less volatile due to intermolecular hydrogen bonding which causes the association of molecules.
APPEARS IN
RELATED QUESTIONS
Write the main product(s) in each of the following reactions:
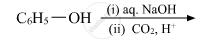
On distilling phenol with Zn dust, one gets:
In the reaction of phenol with CHCl3 and aqueous NaOH at 343 K, the electrophile attacking the ring is:
When Phenol is distilled with zinc dust, it gives:
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why?
Convert the following:
Phenol to N-phenylethanamide.
Which of the following reacts with phenol to give salicylaldehyde after hydrolysis?
Write the chemical equation involved in the following reactions:
Acetylation of salicylic add
Write the name of the reaction, structure and IUPAC name of the product formed when:
Phenol reacts with CHCl3 in the presence of NaOH followed by hydrolysis.
