Advertisements
Advertisements
प्रश्न
The wavelength of Kα X-ray of tungsten is 21.3 pm. It takes 11.3 keV to knock out an electron from the L shell of a tungsten atom. What should be the minimum accelerating voltage across an X-ray tube having tungsten target which allows production of Kα X-ray?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
उत्तर
Given:-
Wavelength of X-ray of tungsten,
`lambda` = 21.3 pm
Energy required to take out electron from the L shell of a tungsten atom, EL = 11.3 keV
Voltage required to take out electron from the L shell of a tungsten atom, VL = 11.3 kV
Let EK and EL be the energies of K and L, respectively.
`E_K - E_L = (hc)/lambda`
Here,
h = Planck's constant
c = Speed of light
`E_K - E_L = (1242 "eV" - "nm")/(21.3 xx 10^-12)`
`E_K - E_L = (1242 xx 10^-9 "eV")/(21.3 xx 10^-12)`
`E_K - E_L = 58.309 "keV"`
`E_L = 11.3 "keV"`
`therefore E_K = 69.609 "keV"`
Thus, the accelerating voltage across an X-ray tube that allows the production of Kα X-ray is given by
VK = 69.609 kV
APPEARS IN
संबंधित प्रश्न
What role dose infra-red radiation play in maintain the Earth’s warmth
What physical quantity is the same for X-rays of wavelength 10−10 m, red light of wavelength 6800 Å and radiowaves of wavelength 500 m?
What is the range of the wavelength of the following electromagnetic waves?
(a)X-rays,
Name the rays or waves of highest frequency .
Name the waves of wavelength nearly 0.1 nm.
Two waves A and B have wavelength 0.01 Å and 9000 Å respectively.
- Name the two waves.
- Compare the speeds of these waves when they travel in vacuum.
The wavelength of X-rays is 0.01 Å. Calculate its frequency. State the assumption made, if any.
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
Why is exposure to X-rays injurious to health but not exposure to visible light, when both are electromagnetic waves?
Find the cutoff wavelength for the continuous X-rays coming from an X-ray tube operating at 30 kV.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The wavelengths of Kα and Lα X-rays of a material are 21.3 pm and 141 pm respectively. Find the wavelength of Kβ X-ray of the material.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the radiations used for the detection of fracture in bones.
Light of wavelength `3500A` is incident on two metals A and B whose work functions are 3.2 eV and 1.9 eV respectively. Which metal will emit photoelectrons?
Ozone layer above the earth's atmosphere will
The half-value thickness of an absorber is defined as the thickness that will reduce exponentially the intensity of a beam of particles by a factor of 2. The half-value thickness in (µm) for lead assuming X-ray beam of wavelength 20 pm, µ = 50 cm-1 for X-rays in lead at wavelength λ = 20 pm, is ______ µm.
Choose the correct option related to wavelengths (λ) of different parts of the electromagnetic spectrum.
Below is an incomplete table showing the arrangement of electromagnetic spectrum in the increasing order of their wavelength. Complete the table:
| Gamma ray | X - ray | UV rays | Visible rays | Infrared | A | Radio waves |
- Identify the radiation A.
- Name the radiation used to detect fracture in bones.
- Name one property common to both A and Radio waves.
What happens when an electron collides with a positron?
Name the electromagnetic radiation that has been used in obtaining the image below.
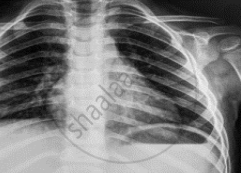
What is the wavelength range of electromagnetic radiation used in radio broadcast?
