Advertisements
Advertisements
प्रश्न
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
उत्तर
Acid dehydration of primary alcohols to ethers occurs by SN2 mechanism in which the nucleophilic attack of the alcohol molecule takes place on the protonated alcohol molecule.
\[\ce{CH3CH2CH2\overset{\bullet\bullet}{\underset{\bullet\bullet}{O}}H + CH3CH2CH2 - \overset{+}{\underset{\bullet\bullet}{O}}\overset{+}{H} ->[S_{N}2][-H^+, -H2O] CH3CH2CH2 - O - CH2CH2CH3}\]
Under these conditions, secondary and tertiary alcohols give alkenes instead of ethers. There is no nucleophilic attack of the alcohol molecule on the protonated alcohol molecule. Instead protonated secondary and tertiary alcohols lose a molecule of water to form stable 2° and 3° carbocations. These carbocations preferentially lose H+ to form alkenes.
\[\begin{array}{cc}
\phantom{.......}\ce{CH3}\phantom{.................}\ce{CH3}\phantom{....................}\ce{CH3}\phantom{.}\\\
\phantom{.....}|\phantom{.....................}|\phantom{........................}|\phantom{..}\\
\ce{\underset{\underset{(2^\circ alcohol)}{Propan-2-ol}}{CH3 - CH - OH} ->[H^-] \underset{Protonated 2^\circ alcohol}{CH3 - CH - \overset{+}{O}H2} ->[][-H2O] \underset{2^\circ Carbocation}{CH3 - CH^+}}
\end{array}\]
\[\begin{array}{cc}
\phantom{}\ce{CH3}\phantom{.......}\ce{CH3}\phantom{.........................}\ce{CH3}\phantom{..................}\\
\phantom{}|\phantom{..........}|\phantom{.............................}|\phantom{....................}\\
\ce{\underset{2-propoxy-2-propane}{CH3 - CH - O - CH - CH3} ->[CH3CHOCH3][-H^+] CH3 - CH^+ ->[][-H^+] \underset{Propene}{CH3 - CH = CH2}}
\end{array}\]
Similarly, 3° alcohols give alkenes instead of ethers.
\[\ce{\underset{\underset{(3^\circ alcohol)}{2-Methylpropan-2-ol}}{(CH3)3C - OH} ->[H^+] \underset{\underset{(3^\circ alcohol)}{Protonated 2-methylpropan-2-ol}}{(CH3)3C - \overset{+}{O}H2} ->[][-H2O] \underset{3^\circ butyl carbocation}{(CH3)3C^+}}\]
\[\begin{array}{cc}
\phantom{...}\ce{CH3}\phantom{.....}\ce{CH3}\phantom{.......................}\ce{CH3}\phantom{............}\ce{CH3}\phantom{...}\\
\phantom{.......}|\phantom{.........}|\phantom{..........................}|\phantom{................}|\phantom{.........}\\
\ce{CH3 - C - O - C - CH3 ->[(CH3)3COH][-H^+] CH3 - C^+ ->[][-H^+] \underset{2-Methylprop-1-ene}{CH3 - CH = CH2}}\\
\phantom{.....}|\phantom{.........}|\phantom{..........................}|\phantom{........................}\\
\phantom{}\ce{\underset{2-Methyl-2-(2-methyl-2-propoxy)propane}{\phantom{..}CH3\phantom{......}CH3}}\phantom{..........}\ce{\underset{3^\circ butyl carbocation}{CH3}}\phantom{....................}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write the reactions of Williamson synthesis of 2-ethoxy-3-methylpentane starting from ethanol and 3-methylpentan-2-ol.
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
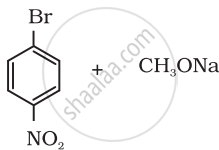 |
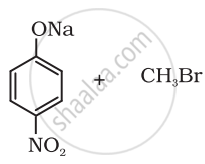 |
| (i) | (ii) |
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of the reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
1-Methoxyethane
Williamson's synthesis of preparing dimethyl ether is a/an ____________.
\[\ce{(CH3)3CONa + CH3CH2Cl ->[-NaCl] (CH3)3COC2H5}\] is called:
Explain why low molecular mass alcohols are soluble in water.
HBr reacts with \[\ce{CH2 = CH - OCH3}\] under anhydrous conditions at room temperature to give ______.
Which of the following reactions are feasible?
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of the reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Short Answer Question.
Identify the product (s) is/are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
