Advertisements
Advertisements
प्रश्न
Prepare a list of objects around you that are electroplated.
उत्तर
Examples of electroplated objects are as follows:
- Chromium plating is done on different parts of cars, buses and motor cycles to give them a shiny appearance.
- A fine layer of gold is deposited on the silver ornaments and they are called gold-plated ornaments.
- Iron used in constructing a building is coated with a layer of zinc. This protects iron from corrosion and rusting.
APPEARS IN
संबंधित प्रश्न
The process of depositing a layer of any desired metal on another material by means of electricity is called ______.
Name three liquids, which when tested in the manner shown in Figure, may cause the magnetic needle to deflect.
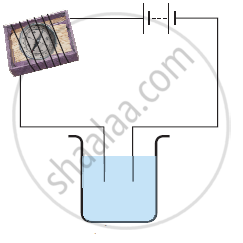
The process that you saw in the image is used for the purification of copper. A thin plate of pure copper and a thick rod of impure copper are used as electrodes. Copper from impure rod is sought to be transferred to the thin copper plate. Which electrode should be attached to the positive terminal of the battery and why?
In an electrolyte the current is due to the flow of ______.
Electroplating is an example for ______.
Why is tin electroplated on iron to make cans used for storing food?
In the circuit given in the figure,
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
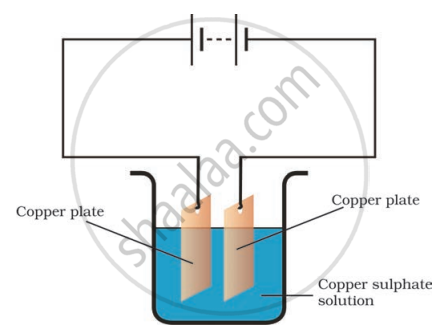
The terminal which is connected to a positive terminal of a battery is called ______.
Assertion: Parts of cars and bicycles are made iron with chromium coating.
Reason: Chromium does not corrode and resist scratches.
What are anodes and cathodes?
