Advertisements
Advertisements
प्रश्न
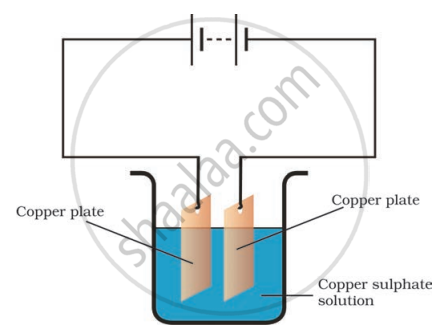
The process that you saw in the image is used for the purification of copper. A thin plate of pure copper and a thick rod of impure copper are used as electrodes. Copper from impure rod is sought to be transferred to the thin copper plate. Which electrode should be attached to the positive terminal of the battery and why?
उत्तर
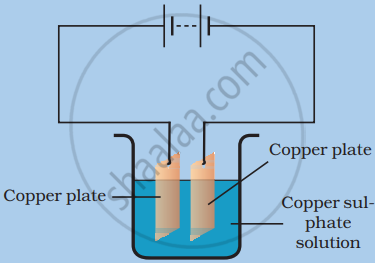
The rod of the impure copper plate should be connected to the battery's positive terminal. Because when an electric current is sent through a copper sulphate solution, it separates into copper and sulphate ions. The free copper is drawn to the battery's negative terminal and deposited on it. On the other hand, the impure copper rod connected to the battery's positive terminal would gain back the copper lost in the solution.
APPEARS IN
संबंधित प्रश्न
Name three liquids, which when tested in the manner shown in Figure, may cause the magnetic needle to deflect.
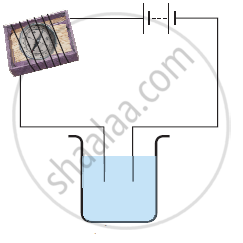
In an electrolyte the current is due to the flow of ______.
An electric current can produce
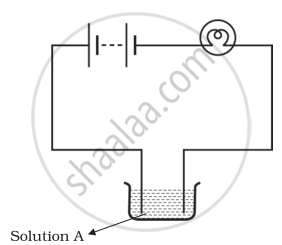
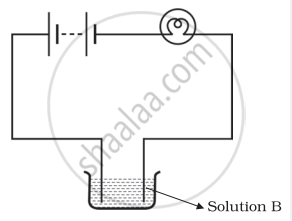
Boojho and Paheli performed experiments taking similar bulbs and cells but two different solutions A and B as shown in the figure.
 |
 |
| Boojho's Experiment | Paheli's Experiment |
| (A) | (B) |
They found that the bulb in the set up A glows more brightly as compared to that of the set up B. You would conclude that
The object to be electroplated is taken as ______ electrode.
In the circuit given in the figure,
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
Explain the following:
Short circuit.
The most common industrial application of chemical effects of electric current is ______.
Liquids that conduct electricity are the solutions of ______.
Name the process which shows the chemical effect of electric current?
