Advertisements
Advertisements
प्रश्न
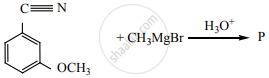
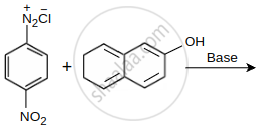
Product ‘P’ in the above reaction is:
विकल्प
उत्तर
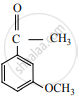
APPEARS IN
संबंधित प्रश्न
The conversion of primary aromatic amines into diazonium salts is known as ___________
Ammonium salt of benzoic acid is heated strongly with P2O5 and the product so formed is reduced and then treated with NaNO2/HCl at low temperature. The final compound formed is ____________.
Identify X in the sequence given below
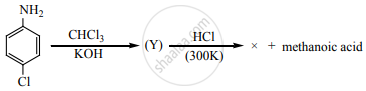
The major product of the following reaction:
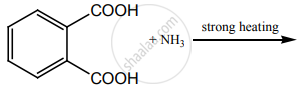
Account for the following.
Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
A dibromo derivative (A) on treatment with KCN followed by acid hydrolysis and heating gives a monobasic acid (B) along with the liberation of CO2. (B) on heating with liquid ammonia followed by treating with Br2/KOH gives (C) which on treating with NaNO2 and HCl at low temperature followed by oxidation gives a monobasic acid (D) having molecular mass 74. Identify A to D.
Why is \[\ce{NH2}\] group of aniline acetylated before carrying out nitration?
What would be the major product of the following reaction?
\[\ce{C6H5 - CH2 - OC6H5 + HBr -> A + B}\]
Coupling of benzene diazonium chloride with 1-naphthol in alkaline medium will give:
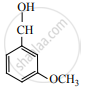
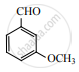
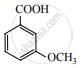
The major product of the following reaction is: