Advertisements
Advertisements
प्रश्न
Show the formation of Na2O by the transfer of electrons.
उत्तर
`Na_2O`
Na=11=2,8,1 (Valency = 1)
O = 8 = 2,6 (Valency = 2)
(a) `Na → Na^(+)+ e^-`
`(Na → Na^(+)+e^-) xx 2`
`2Na → 2Na + 2e^-`
(b) `O + e^(-)→ O^-`
`(O + e^(-) → O^- ) xx 2`
`2O + 2e^(-) → O^-`
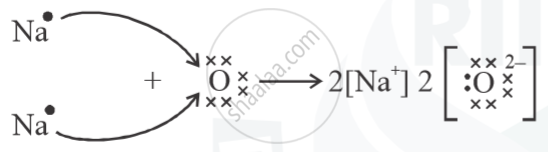
`i.e. 2Na + O → Na_2O`
APPEARS IN
संबंधित प्रश्न
Balance the given equation:
HNO3 + Ca(OH)2  Ca(NO3)2 + H2O
Ca(NO3)2 + H2O
A metal X forms a salt XSO4. The salt XSO4 forms a clear solution in water which reacts with sodium hydroxide solution to form a blue precipitate Y. Metal X is used in making electric wire and alloys like brass.
(a) What do you think metal X could be?
(b) Write the name, formula and colour of salt XSO4.
(c) What is the blue precipitate Y?
(d) Write a chemical equation of the reaction which takes place when salt XSO4 reacts with sodium hydroxide solution. Give the state symbols of all the reactants and products which occur in the above equation.
Balance the following chemical equation :
H2 + O2 → H2O
Write your observation and name the product when ron nails are added to an aqueous solution of copper sulphate.
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Write the balanced chemical equations of the following reactions.
chlorine + sulphur dioxide + water → sulphuric acid + hydrogen chloride
Write the balanced chemical equation of the following reaction.
zinc sulphide + oxygen → zinc oxide + sulphur dioxide
Write word equation for the following molecular equation:
\[\ce{N2 + 3H2 ⇌[Fe - 450°C][200 atoms] 2NH3 + \triangle}\]
Word equation:
State why [+ Δ] indication is seen after NH3.
State what 200 atmospheres – indicates.
State the function of Fe in the above reaction.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – copper carbonate.
Write a balanced equation for the following word equation:
Potassium bicarbonate → Potassium carbonate + Water + Carbon dioxide
