Advertisements
Advertisements
Question
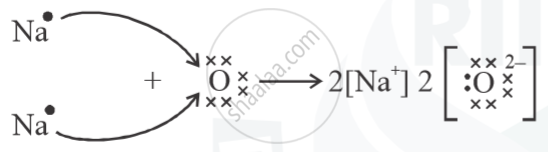
Show the formation of Na2O by the transfer of electrons.
Solution
Na=11=2,8,1 (Valency = 1)
O = 8 = 2,6 (Valency = 2)
(a)
(b)
APPEARS IN
RELATED QUESTIONS
Translate the following statement into chemical equation and then balance it.
Hydrogen sulphide gas burns in the air to give water and sulphur dioxide.
Find the odd man out:
Turmeric, Methyl Orange, Rose petals, Beetroot.
What is a chemical equation? Explain with the help of an example.
Which of the following does not involve a chemical reaction?
Write balanced chemical equation with state symbols for the following reaction:
Barium chloride solution reacts with sodium sulphate solution to give insoluble barium sulphate and a solution of sodium chloride.
Name the type of chemical reaction shown by the following equation:
2Mg+ O2 → 2MgO
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of acid required.
Balance the following simple equation:
Mg + CO2 → MgO + C
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
PbO2 +4HCl → PbCl2 + H2O + Cl2
The chemical equation Na2SO4(aq) + BaCl2(aq) → BaSO4(s)↓ + 2NaCl(aq) represents which of the following types of reaction?
