Advertisements
Advertisements
Question
The chemical equation Na2SO4(aq) + BaCl2(aq) → BaSO4(s)↓ + 2NaCl(aq) represents which of the following types of reaction?
Options
Neutralization
Combustion
Precipitation
Single displacement
Solution
Precipitation
APPEARS IN
RELATED QUESTIONS
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are compounds.
Give one example of an endothermic reaction.
Balance the following chemical equation:
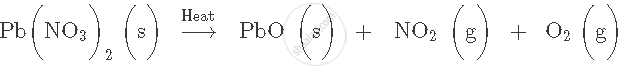
Express the following facts in the form of a balanced chemical equation:
"When a strip of copper metal is placed in a solution of silver nitrate, metallic silver is precipitated and a solution containing copper nitrate is formed".
Write symbolic representation for the following word equation and balance them :
Carbon + Oxygen → Carbon dioxide.
Write word equation for the following molecular equation:
\[\ce{CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2↓}\]
Word equation:
State the colour of the products.
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{Na2SO3 + 2HCl ->[\triangle] 2NaCl + H2O + SO2}\]
CaCO3 + 2HCl[dil.] → CaCl2 + H2O + CO2 [g]
State the information not conveyed by the above chemical equation.
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Balance the equation with proper coefficient and rewrite the equation.
Dil. HCl is added to Zn granules. How will you prove that chemical change has taken place here? Support your response with two arguments.
