Advertisements
Advertisements
प्रश्न
Some metals react with acids to produce salt and hydrogen gas. Illustrate it with an example. How will you test the presence of this gas?
उत्तर
Some metals react with acids to produce salt and hydrogen gas. For example, when magnesium reacts with hydrochloric acid, magnesium chloride (a salt) and hydrogen gas are produced. The chemical equation for this reaction is:
\[\ce{Mg + 2HCL -> MgCl2 + H2}\]
To test for the presence of hydrogen gas, bring a burning match or candle near the mouth of the reacting test tube. If hydrogen gas is present, it will ignite with a 'pop' sound, characteristic of hydrogen gas. This is because hydrogen gas is highly flammable and reacts with oxygen in the air to produce water vapour when ignited, making a popping sound.
APPEARS IN
संबंधित प्रश्न
Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
The litmus paper or the litmus solution is obtained from ________ plant
(A) Moss
(B) Lichen
(C) Rose
(D) Hibiscus
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
wasp stings?
Phenolphthalein is a synthetic type of indicator.
Egg shell is made up of ____________.
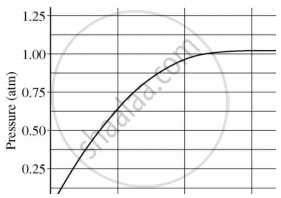
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to ______
Which of the following is used for dissolution of gold?
“All acids contain one or more hydrogen atoms, but not all substances containing hydrogen are acids.” Support this statement with examples.
What property do acids and bases have in common? Explain it with an example.
