Advertisements
Advertisements
प्रश्न
State and explain Hess’s law of constant heat summation.
उत्तर
- Hess's law of constant heat summation states that," The change in enthalpy for a reaction is the same whether the reaction takes place in one or a series of steps".
- The Hess's law is a direct consequence of fact that the enthalpy is a state function and so enthalpy change depends only on the initial and final states of the system and not on the path by which the reaction takes place.
eg. The conversion of A to C can take place directly
in a single step.
A → C, ΔH° = ΔH1 - The reaction can also take place in two steps for which ΔH°
values are known.
Step (1) A → B, ΔH° = ΔH2
Step (2) B → C, ΔH° = ΔH3
____________________________
Overall A → C, ΔH° = ΔH2 + ΔH3 - According to Hess's law, ΔH1 = ΔH2 + ΔH3.
The sequence of steps is represented in the figure.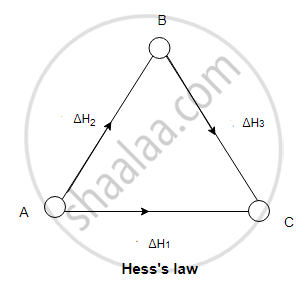
APPEARS IN
संबंधित प्रश्न
State and explain Hess’s law of constant heat summation.
Enthalpy is an extensive property. In general, if enthalpy of an overall reaction A→B along one route is ∆rH and ∆rH1, ∆rH2, ∆rH3 ..... represent enthalpies of intermediate reactions leading to product B. What will be the relation between ∆rH for overall reaction and ∆rH1, ∆rH2 ..... etc. for intermediate reactions.
The difference between CP and CV can be derived using the empirical relation H = U + pV. Calculate the difference between CP and CV for 10 moles of an ideal gas.
The enthalpy of vapourisation of \[\ce{CCl4}\] is 30.5 kJ mol–1. Calculate the heat required for the vapourisation of 284 g of \[\ce{CCl4}\] at constant pressure. (Molar mass of \[\ce{CCl4}\] = 154 g mol–1).
Given that:
\[\ce{C + O2 -> CO2; ΔH° = - x {k}J}\]
\[\ce{2CO + O2 -> 2CO2; ΔH° = - y {k}J}\]
The heat of formation of carbon monoxide will be ______.
