Advertisements
Advertisements
प्रश्न
State one use of radio-isotopes.
उत्तर
The most important one use of radio-isotopes is in medicines which are used mainly to cure cancer.
APPEARS IN
संबंधित प्रश्न
Why do Free electrons not leave the metal surface on their own
A nucleus `""_11^24Na` emits a beta particle to change into Magnesium (Mg)
(i) Write the symbolic equation for the process.
(ii) What are numbers 24 and 11 called?
(iii) What is the general name of `""_12^24Mg `with respect to `""_11^24Na` ?
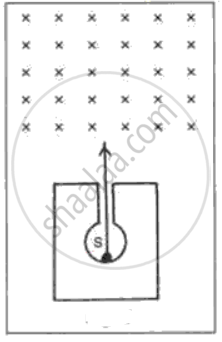
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
A radioactive nucleus `""_"Z"^"A"` X first emits a beta particle and then an alpha particle to give the resulting nucleus `""_"Q"^"P"` Y What will be the values of P and Q in terms of A and Z?
State any two uses of cathode rays.
In a cathode ray tube state the purpose of covering cathode by thorium and carbon.
A certain nucleus has a mass number 20 and atomic number 9. Find the number of neutrons and protons present in it.
Which of the following statements is/are correct?
- α particles are photons
- Penetrating power of γ radiation is very low
- Ionization power is maximum for α rays
- Penetrating power of γ radiation is very high
A radioactive nucleus \[\ce{^A_ZX}\] undergoes spontaneous decay in the sequence \[\ce{^A_ZX -> _{Z-1 }B -> _{Z-3}C -> _{Z-2}D }\], where Z is the atomic number of element X. The possible decay particles in the sequence are ______.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
