Advertisements
Advertisements
प्रश्न
A nucleus `""_11^24Na` emits a beta particle to change into Magnesium (Mg)
(i) Write the symbolic equation for the process.
(ii) What are numbers 24 and 11 called?
(iii) What is the general name of `""_12^24Mg `with respect to `""_11^24Na` ?
उत्तर
The `""_11^24Na` nucleus emits a beta particle and changes into Magnesium.
(i) The above process is represented by the equation
`""_11^24Na ->""_12^24Mg + ""_-1^0e`
sodium Magnesium Beta particle
(ii) The number 24 is the mass number of the nucleus and 11 is the atomic number.
(iii) `""_11^24Na `and `""_12^24Mg ` are isobars, i.e., `""_12^24Mg ` is an isobar of `""_11^24Na ` as they have same mass number (protons + neutrons) but different atomic number (protons).
APPEARS IN
संबंधित प्रश्न
Write one use of cathode ray tube.
Represent the change in the nucleus of a radioactive element when a β particle is emitted.
What happens if the negative potential is changed on a grid?
Radiations given out from a source when subjected to an electric field in a direction perpendicular to their path are shown below in the diagram. The arrows show the path of the radiation A, B and C. Answer the following questions in terms of A, B and C.
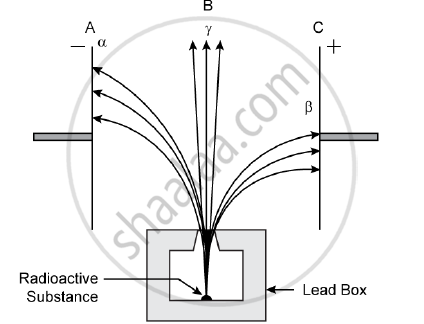
1) Name the radiation B which is unaffected by the electrostatic field.
2) Why does the radiation C deflect more than A?
3) Which among the three causes the least biological damage externally.
4) Name the radiation which is used in carbon dating.
What is meant by radioactivity?
What do you mean by Atomic number
What are the uses of radioactive isotopes in following?
(a) Medical field,
(b) Agriculture,
(c) Industries?
A radioactive source emits three types of radiations.
Name the radiation which has the highest ionizing power.
State one use of radio-isotopes.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
