Advertisements
Advertisements
प्रश्न
State three properties of ultra-violet radiation similar to visible light.
उत्तर
(i) Ultra-violet radiations can travel in vacuum as the visible light can do, with the speed 3 × 108 ms−1.
(ii) Both the ultra-violet radiations and the visible light are electromagnetic waves.
(iii) Both the ultra-violet radiations and the visible light obey the laws of reflection and refraction.
APPEARS IN
संबंधित प्रश्न
To which part of the electromagnetic spectrum does a wave of frequency 5 × 1019 Hz belong?
Name the subjective property of light related to its wavelength.
A wave has a wavelength of 10-3 nm. Name the wave.
Name the rays or waves of highest frequency .
50% of the X-ray coming from a Coolidge tube is able to pass through a 0.1 mm thick aluminium foil. The potential difference between the target and the filament is increased. The thickness of the aluminium foil that will allow 50% of the X-ray to pass through will be
Mark the correct options.
(a) An atom with a vacancy has smaller energy that a neutral atom.
(b) K X-ray is emitted when a hole makes a jump from the K shell to some other shell.
(c) The wavelength of K X-ray is smaller than the wavelength of L X-ray of the same material.
(d) The wavelength of Kα X-ray is smaller than the wavelength of Kβ X-ray of the same material.
Iron emits Kα X-ray of energy 6.4 keV. Calculate the times taken by an iron Kα photon to cross through a distance of 3 km.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
If the operating potential in an X-ray tube is increased by 1%, by what percentage does the cutoff wavelength decrease?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
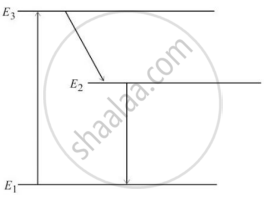
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
