Advertisements
Advertisements
प्रश्न
The correct order of O – O bond length in hydrogen peroxide, ozone and oxygen is
विकल्प
H2O2 > O3 > O2
O2 > O3 > H2O2
O2 > H2O2 > O3
O3 > O2 > H2O2
उत्तर
O2 > O3 > H2O2
APPEARS IN
संबंधित प्रश्न
Give a reason for carbon is tetravalent in nature.
Identify the type of overlap present in H2. Explain diagrammatically.
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule?
Among the following, the compound that contains, ionic, covalent and Coordinate linkage is ______.
What is a pi - bond?
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2py
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2py
Why does type of overlap given in the following figure not result in bond formation?
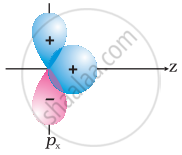 |
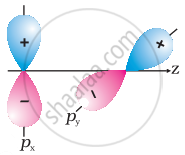 |
Match List - I with List - II.
| List - I | List - II | ||
| (a) | \[\ce{PCl5}\] | (i) | Square pyramidal |
| (b) | \[\ce{SF6}\] | (ii) | Trigonal planar |
| (c) | \[\ce{BrF5}\] | (iii) | Octahedral |
| (d) | \[\ce{BF3}\] | (iv) | Trigonal bipyramidal |
Choose the correct answer from the options given below.
The \[\ce{H - N - H}\] bond angle in ammonia molecule is ______.
