Advertisements
Advertisements
प्रश्न
What is a pi - bond?
उत्तर
When two atomic orbitals overlap sideways, the resultant covalent bond is called a pi (π) bond. When we consider x-axis as the molecular axis, the py – py and pz – pz overlaps will result in the formation of a π – bond.
APPEARS IN
संबंधित प्रश्न
Draw diagram for bonding in ethene with sp2 Hybridisation.
Display electron distribution around the oxygen atom in the water molecule and state the shape of the molecule, also write the H-O-H bond angle.
Complete the following Table.
| Molecule | Type of Hybridization | Type of bonds | Geometry | Bond angle |
| CH4 | - | 4C-H 4σ bonds |
Tetrahedral | - |
| NH3 | sp3 | 3N-H 3σ bonds 1 lone pair |
- | - |
| H2O | - | - | angular | 104.5° |
| BF3 | sp2 | - | - | 120° |
| C2H4 | - | - | - | 120° |
| BeF2 | - | 2 Be-F | Linear | - |
| C2H2 | sp | (3σ+2π) 1C-C σ 2C-H σ 2C-C π |
- | - |
The ratio of number of sigma (σ) and pi (л) bonds in 2- butynal is ______.
In ClF3, NF3 and BF3 molecules the chlorine, nitrogen and boron atoms are ______.
Among the following, the compound that contains, ionic, covalent and Coordinate linkage is ______.
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2py
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2pz
The number of sigma bonds in paracetamol is ____________.
Why does type of overlap given in the following figure not result in bond formation?
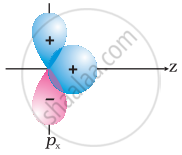 |
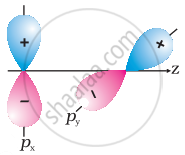 |
