Advertisements
Advertisements
Question
What is a pi - bond?
Solution
When two atomic orbitals overlap sideways, the resultant covalent bond is called a pi (π) bond. When we consider x-axis as the molecular axis, the py – py and pz – pz overlaps will result in the formation of a π – bond.
APPEARS IN
RELATED QUESTIONS
Display electron distribution around the oxygen atom in the water molecule and state the shape of the molecule, also write the H-O-H bond angle.
Give a reason for the sigma (σ) bond is stronger than the Pi (π) bond.
Complete the following Table.
| Molecule | Type of Hybridization | Type of bonds | Geometry | Bond angle |
| CH4 | - | 4C-H 4σ bonds |
Tetrahedral | - |
| NH3 | sp3 | 3N-H 3σ bonds 1 lone pair |
- | - |
| H2O | - | - | angular | 104.5° |
| BF3 | sp2 | - | - | 120° |
| C2H4 | - | - | - | 120° |
| BeF2 | - | 2 Be-F | Linear | - |
| C2H2 | sp | (3σ+2π) 1C-C σ 2C-H σ 2C-C π |
- | - |
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule?
The correct order of O – O bond length in hydrogen peroxide, ozone and oxygen is
Among the following, the compound that contains, ionic, covalent and Coordinate linkage is ______.
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2pz
Which of the following is correct decreasing order of the repulsive Interaction of electron pairs in a molecule?
Why does type of overlap given in the following figure not result in bond formation?
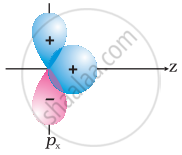 |
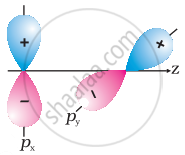 |
Which of the following statements is INCORRECT according to the valence bond theory?
