Advertisements
Advertisements
Question
Define σ – bond.
Solution
When two atomic orbitals overlap linearly along the axis, the resultant bond is called a sigma (σ) bond.
APPEARS IN
RELATED QUESTIONS
Draw an orbital diagram of Fluorine molecule
Give reasons for need of Hybridization
Identify the type of overlap present in H2. Explain diagrammatically.
Mention the steps involved in Hybridization.
In ClF3, NF3 and BF3 molecules the chlorine, nitrogen and boron atoms are ______.
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2pz
The overlap of orbitals involved in the formation of C - Br bond in vinyl bromide is ______.
Why does type of overlap given in the following figure not result in bond formation?
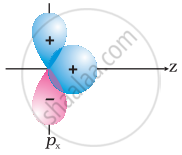 |
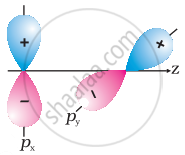 |
Match List - I with List - II.
| List - I | List - II | ||
| (a) | \[\ce{PCl5}\] | (i) | Square pyramidal |
| (b) | \[\ce{SF6}\] | (ii) | Trigonal planar |
| (c) | \[\ce{BrF5}\] | (iii) | Octahedral |
| (d) | \[\ce{BF3}\] | (iv) | Trigonal bipyramidal |
Choose the correct answer from the options given below.
The \[\ce{H - N - H}\] bond angle in ammonia molecule is ______.
