Advertisements
Advertisements
प्रश्न
The correct order of the increasing basic nature of Ammonia, Methylamine and Aniline is:
विकल्प
Methylamine < Aniline < Ammonia
Aniline < Methylamine < Ammonia
Ammonia < Aniline < Methylamine
Aniline < Ammonia < Methylamine
उत्तर
Aniline < Ammonia < Methylamine
APPEARS IN
संबंधित प्रश्न
Why does NH3 act as a Lewis base?
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2
Write the structures of the main products of the following reactions:
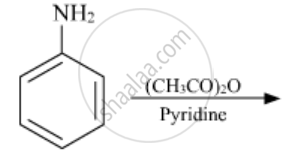
The following reaction takes place in the presence of:
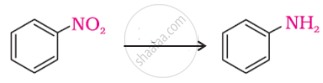
The most reactive amine towards dilute hydrochloric acid is:
The correct decreasing order of basic strength of the following species is ______.
\[\ce{H2O, NH3, OH-, NH^{-}2}\]
When methyl iodide is heated with ammonia, what is the product obtained?
Which of the following is most basic?
What is the characteristic smell of liquid amines?
Give reasons for the following observation:
pKb of aniline is lower than the m-nitroaniline.
