Advertisements
Advertisements
प्रश्न
The following graph represents the pressure versus number density for an ideal gas at two different temperatures T1 and T2. The graph implies
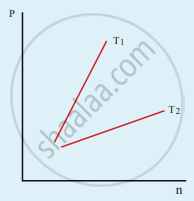
विकल्प
T1 = T2
T1 > T2
T1 < T2
Cannot be determined
उत्तर
T1 > T2
APPEARS IN
संबंधित प्रश्न
Find the kinetic energy of 3 litre of gas at S.T.P given standard pressure is 1.013 × 105 N/m2.
A sample of an ideal gas is at equilibrium. Which of the following quantity is zero?
Two identically sized rooms A and B are connected by an open door. If the room A is air-conditioned such that its temperature is 4°C lesser than room B, which room has more air in it?
The ratio γ = `"C"_"p"/"C"_"v"` for a gas mixture consisting of 8 g of helium and 16 g of oxygen is ____________.
What is the microscopic origin of pressure?
Estimate the total number of air molecules in a room of a capacity of 25 m3 at a temperature of 27°C.
A perfect gas of 'N' molecules, each of mass 'm', moving with velocities 'C1', 'C2', ...... .'CN' is enclosed in a cubical vessel of volume 'V'. The pressure exerted by the gas on the walls of the vessel is ______. ('p' = density of gas)
The average force applied on the walls of a closed container depends as 'Tx', where 'T' is the temperature of an ideal gas. The value of 'x' is ______.
The kinetic energy per molecule of a gas at temperature T is ______.
The velocities of five molecules are 2 m/s, 3 m/s, 4 m/s, 5 m/s and 6 m/s. Find the root mean square velocity of molecules.
