Advertisements
Advertisements
प्रश्न
The half-life of \[\ce{_38^90Sr}\] is 28 years. Determine the disintegration rate of its 5 mg sample.
उत्तर
Data: T1/2 = 28 years = 28 x 3.156 x 107 s
= 8.837 x 108 s, M = 5mg = 5 x 10−3 g
90 grams of \[\ce{_38^90Sr}\] contain 6.02 x 1023 atoms
Hence, here, N = `((6.02 xx 10^23)(5 xx 10^-3))/90`
= 3.344 × 1019 atoms
∴ The disintegration rate = `"N"lambda = "N"0.693/("T"_(1//2))`
`= ((3.344 xx 10^19)(0.693))/(8.837 xx 10^8)`
= 2.622 x 1010s−1
APPEARS IN
संबंधित प्रश्न
What is the difference between a nuclear reactor and a nuclear bomb?
Determine the maximum angular speed of an electron moving in a stable orbit around the nucleus of the hydrogen atom.
The mass of the earth is 81 times the mass of the moon and the distance between their centres is R. The distance from the centre of the earth, where gravitational force will be zero is ______.
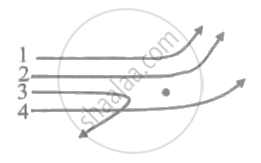
The diagram shows the path of four `alpha` - particles of the same energy being scattered by the nucleus of an atom simultaneously. Which of these are/is not physically possible?
Two protons are kept at a separation of 10 nm. Let Fn and Fe be the nuclear force and the electromagnetic force between them ____________.
The radius of germanium (Ge) nuclide is measured to be twice the radius of `""^9""_4"Be"`. The number of nucleons in Ge will be ____________.
If radius of the `{:(27),(13):}` Al nucleus is taken to be RAI then the radius of `{:(125), (53):}` Te nucleus is ____________.
A sample of radioactive element contains 8 × 1016 active nuclei. The half-life of the element is 15 days. The number of nuclei decayed after 60 days is ______.
Explain how nuclear size of an atom is estimated.
Define the term Nucleons.
